 |
  AIDS/HIV AIDS/HIV
Role of the Thymus in T Cell Homeostasis
By monitoring the changes in phenotypic T cell markers as well as in the numbers of T cell Receptor (TCR) excisional circles – a recently described marker for recent thymic emigrants – following thymectomy, we have found evidence that surgical thymectomy in juvenile macaques results in a faster decay of peripheral CD4+ T cells, but does not cause a substantial shift in CD45RA+ (naive) and CD45RA- (memory) populations. No compensatory extra-thymic source was detected in lymphoid tissues, although there was a small compensatory increase in T cell proliferation in the peripheral T cell pool. After SIV infection, thymectomized animals did not have higher viral loads, greater T cell decay, or faster disease progression. We therefore conclude that peripheral destructive processes, rather than a loss of thymic output, appear to be the main causes of T cell depletion in SIV infection. Aaron Diamond AIDS Research Center.
Microbicides to Prevent AIDS Transmission
Cellulose acetate phthalate (CAP), a pharmaceutical excipient designed as a coating material for tablets or granules, has been demonstrated to be effective against herpes simplex virus type 2 (HSV-2) infection in mice, and to protect four of six rhesus monkeys from vaginal challenge with simian immunodeficiency virus SIVmac251. To assess whether CAP confers protection against primary viral strains that are transmitted in humans, infections with pathogenic simian/human immunodeficiency viruses (SHIVs) expressing the envelopes of X4 and R5HIV-1 strains (SHIVSF33A and SHSIVSF162P3, respectively) were performed. Seven of ten CAP treated macaques were protected from challenge with a mixture of X4-SHIVSF33A and R5-SHIVSF162P3. These findings in macaques suggest that CAP is efficacious against both X4 and R5 SHIV viruses in vivo, and should therefore be considered as a viable topical microbicide candidate in the prevention of HIV-1 infection. Aaron Diamond AIDS Research Center.
SIV Epitope Specific CTL Responses in MAMU-A*01A*02 Double-positive Macaques
A vaccine-elicited cytotoxic T-cell lymphocyte (CTL) response with a maximal breadth of epitope specificities should mediate the most efficient control of HIV-1 replication following infection. We studied the evolution of epitope-specific immune responses in naïve rhesus monkeys following infection with SIVmac251. The delay in the peak Mamu-A*02/pl99RY-specific CTL response in the presence of highly immunodominant Mamu-A*01-restricted CTL responses is most likely due to immunodomination, and represents the first demonstration of immunodomination in outbred primate species. Beth Israel Deaconess Medical Hospital, Harvard Medical School.
AT-2 SIV Vaccination after Dendritic Cell Mobilization
While both immature and mature monocytes-derived dendritic cells capture and present AT-2SIV to primed T cells in vitro, mature dendritic cells activate both CD4 and CD8 T cells but immature dendritic cells predominantly activated CD4 T cells. This supports the rationale for targeting activated dendritic cells with antigen for induction of the most effective immunity. Peripheral dendritic cells and mucosal dendritic cells respond to various stimuli comparable to human dendritic cells and can be mobilized in vivo following treatment with Flt3L. As little as 7 days treatment significantly increases dendritic cell numbers. The peak of the increase in dendritic cell numbers post treatment is under investigation. A study applying AT-2 SIV to the tonsils of naïve macaques revealed that SIV-specific T cells are primed and that pretreatment with immuno-stimulatory oligodeoxynucleotides (ISS-ODNs that activate peripheral dendritic cells), tends to increase the responses seen. Population Council, New York.
Division of Comparative Pathology
More people die from AIDS each year than from any other infectious disease. Over 5 million people die from AIDS every year (about 14,000 per day) making this perhaps the greatest infectious disease threat to the human race. In the United States, AIDS is the second leading cause of death in young adults (18-40 years of age) and is second only to automobile accidents. Investigation of AIDS pathogenesis, prevention and treatment represent the largest research objective of the Division and encompass a number of projects examining 1) the macaque model of neuroAIDS, 2) AIDS and the mucosal immune system, 3) the development of microbicides to prevent HIV heterosexual transmission of AIDS, and 4) testing new therapies for AIDS.
Macaque model of neuroAIDS
Infection of rhesus macaques with SIV results in rapid neuroinvasion and neuropathologic abnormalities that are similar to those observed in HIV-infected humans. We have used this model to examine the role of chemokines and their receptors in neuroinvasion and the development of neuronal injury and neurologic disease. We previously demonstrated elevated expression of the chemokines MIP-1a, MIP-1b, and RANTES and the corresponding receptors CCR3, CCR5, and also CXCR4 in perivascular infiltrates in the brain. Of additional interest was the presence of CCR3, CCR5, and CXCR4 on a subpopulation of large hippocampal and neocortical pyramidal neurons and on glial cells in both normal and SIV-infected brain. The expression of known HIV/SIV coreceptors on neurons and astrocytes suggested a possible mechanism whereby HIV/SIV or the chemokines they induce could interact with these cells, disrupting their normal physiologic function and contributing to the neuropathogenesis of AIDS. To address this hypothesis, we have used immediately ex vivo and in vitro techniques to confirm the presence of all three of these chemokine receptors on a subpopulation of neurons and CCR5 and CXCR4 expression on the majority of astrocytes. These chemokine receptors are functional as demonstrated by increased intracellular calcium in response to the appropriate ligand. We have also used proton magnetic resonance spectroscopy (MRS) to evaluate the neuronal marker n-acetylaspartate (NAA) both in vivo and ex vivo. A 25% decrease in NAA was detected 14 days after infection coincident with peak viremia, neuroinvasion and increased numbers of perivascular macrophages. Decreases in NAA were correlated with decreases in synaptophysin and calbindin in adjacent sections of brain. A further 10% decrease in NAA levels was observed in animals infected for two years or more regardless of the presence of SIV-encephalitis (SIVE). These results indicate that neuronal injury occurs early after viral infection, is associated with neuroinvasion and progresses during the course of infection. The interaction of chemokines or viral envelope with these functional chemokine receptors on neurons and astrocytes suggests a physiologic mechanism whereby neuronal injury could occur.
AIDS and the mucosal immune system
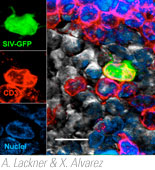
The Division has several ongoing AIDS-related projects to examine the pathogenesis, prevention, and treatment of HIV infection. Much of this work focuses on the vaginal and intestinal mucosa as well as the mucosal immune system in general. Previously, we had shown that rapid and profound CD4+ T cell depletion occurs almost exclusively within the intestinal tract of simian immunodeficiency virus (SIV)-infected macaques within days of infection. In more recent work, we have shown that intestinal CD4+ T cells have much higher expression of CCR5 than T cells in peripheral blood or lymph nodes. Furthermore, the selectivity and extent of CD4+ T cell loss may depend upon these cells co-expressing CCR5 and having a “memory” phenotype. These seminal studies we originally performed in macaques were finally confirmed in HIV-infected humans in 2004 which has resulted in a heightened interest in studying the mucosal immune system and AIDS.
In addition, we have also demonstrated that CCR5 expression on CD4+ T cells may be fundamentally involved in the pathogenesis of AIDS and disease progression (or the lack thereof) by examining expression of these markers in sooty mangabeys (SM) and African green monkeys (AGM). Sooty mangabeys and AGM are the natural host of SIV, and despite persistent high viral loads, these animals rarely progress to AIDS. We have recently discovered that both sooty mangabeys and AGMs naturally have markedly lower percentages of CD4+CCR5+ T cells in their tissues, which could explain why these animals do not progress to AIDS. This finding could have significance for treatment and vaccine strategies in HIV patients.
Heterosexual transmission of AIDS: Development of microbicides to prevent transmission
The development of a microbicide that could be applied to the vagina and prevent the transmission of HIV-1 could save millions of lives. Unfortunately, compounds that destroy HIV-1 are also likely to damage mucosal tissues. Alternatively, fusion inhibitors that attach to viral or host cell receptors may provide a safe and effective mechanism to prevent HIV-1 infection. In 2003, we demonstrated that applying a compound to the vagina of monkeys that blocked the molecule responsible for viral attachment to the host CD4 molecule could completely prevent the vaginal transmission of SHIV (a virus having the HIV outer envelope). In 2004, we demonstrated that a CCR5 blocking agent (PSC-RANTES) could also completely prevent the vaginal transmission of SHIV when introduced as a topical application in the vagina. These studies were the first to demonstrate that CD4 or CCR5 fusion inhibitors could be part of an effective HIV preventative (microbicide). In more recent studies, we have demonstrated that a small molecule inhibitor of CCR5 can prevent vaginal SDHIV transmission. We are continuing to test this fusion inhibitor with the goal of producing a cheap and effective microbicide that could be distributed to places where the epidemic is rampant to slow the spread of HIV infection.
Testing new therapies for AIDS
Although remarkable progress has been made in anti-viral therapies in the last few years, no patient has been cured of infection, and increasingly, patients are showing resistance to anti-HIV drugs in use today. With the rapid and spreading emergence of multi-drug resistant strains of HIV, new classes of anti-HIV therapies are needed to combat HIV infections and for the prevention of AIDS. We are currently testing fusion inhibitors in SIV-infected rhesus macaques to determine whether they may affect viral loads and whether they may be useful as single or combinational therapies against SIV and SHIV-infected macaques. We have demonstrated that certain compounds have remarkable efficacy in reducing viral loads in SIV-infected macaques and are currently testing whether these changes correlate with reductions in chemokine receptor expression, and/or result in viral envelope mutations that may result in drug resistance. These compounds are being tested for efficacy, drug resistance and safety in nonhuman primates.
Division of Immunology
We study virus-specific cellular immune responses specific for the AIDS virus in a nonhuman primate model of AIDS, the simian immunodeficiency virus-infected rhesus monkey. These studies are done using both functional assays and flow cytometry, employing tetrameric MHC class I or II/peptide complexes. These tetramers are used as staining reagents in flow cytometric analyses to quantitate and define the phenotype of antigen-specific T lymphocytes. Using these reagents, we were able to demonstrate a clear correlation between cytotoxic T cell (CTL) expansion and clearance of viral replication during primary SIV infection. Moreover, the distribution of virus-specific CD8+ T cells in various lymphoid compartments and their association with localized virus replication has also been assessed. The precise quantitative power of this technology has allowed us to compare different vaccination modalities in order to develop an effective vaccine against HIV infection. We have also succeeded in developing tetrameric MHC class II peptide complexes to quantitate antigen-specific CD4+ T cells in virus-infected and vaccinated rhesus monkeys. We have selected the rhesus MHC class II, Mamu-DR*W201 for the construction of the class II tetramers, because we found that this molecule is expressed in 35-40% of all rhesus monkeys tested from four different colonies. This new technology has facilitated the quantitative analysis of simian immunodeficiency virus (SIV)-specific T cell responses in vivo in monkeys following virus infection and immunization. To better understand the biology of the antigen–specific CD8+ and CD4+ T lymphocytes in SIV infection, we are currently extending the usage of the tetramer technology to other new MHC class I and II alleles. Recently, to expand our ability to analyze the MHC-TCR interaction from the opposite perspective, we have constructed a TCR tetramer by reassembling a and b chains derived from a peptide-specific T cell population into a functional antigen-specific TCR. The TCR tetramer recognized the restricting MHC class I molecule only in the peptide-bound state and with high specificity and avidity. Importantly, subtle changes in cognate peptide levels bound to the class I molecule were accurately reflected by parallel changes in the mean fluorescence intensity of cells that bound the TCR tetramer. Furthermore, we showed that the TCR tetramer blocks expansion in vitro of CTL specific for the cognate peptide, but has no effect on the expansion of CTL specific for control peptides restricted by the same class I allele. The stringency of this tetramer for its restricting class I allele was also demonstrated by accurately identifying animals expressing the restricting Mamu-A*01 class I allele from a large cohort of over 100 outbred rhesus macaques. With its high throughput potential and reproducibility, TCR tetramer-based approaches can help explore the MHC/peptide interface in detail. With the described tools in hand, we are currently using both mouse and rhesus macaque models to study the fate of memory as well as naïve antigen specific CD8+ and CD4+ T cells using dendritic cells as professional antigen presenting cells. Detailed knowledge of the interaction of APC with antigen specific T cells will be needed to design effective vaccine strategies as well as immune-based therapy to infectious diseases, tumors and autoimmune diseases.
Division of Microbiology
Origins and Genetic Diversity of HIV and the Emergence of Pandemic HIV
Research in the Division has focused on identifying SIV genetic lineages that are ancestral to HIV with the goal of understanding the genetic diversity of primate lentiviruses. Scientists in the Division have discovered new SIVs in western and equatorial Africa that are members of the HIV family tree. The ancestral viruses to HIV-1 and HIV-2 are SIV from the chimpanzee and the sooty mangabey, respectively. However, the SIV group infects over 40 monkey species in Africa. One focus is finding out if the other SIVs that infect other African monkey and ape species pose a risk for human infection and the emergence of new HIV strains and types. Another major effort is to understand the steps leading to the transition of SIV to HIV.
Pathogenesis of SIV
The SIV animal model is being used to conduct basic research on the immunopathogenesis of SIV infections. The research investigates cross species transmission of SIV, long term non progression and the absence of disease in African nonhuman primates naturally infected with SIV.
It is now clear that AIDS originated from cross species transmission of SIV from one or more species of African nonhuman primates to humans. Thus, a better understanding of cross species transmission, emergence, and adaptation of SIV to new human and nonhuman primate hosts is of major importance. The mechanisms by which SIV successfully adapts to new hosts will shed light on the origins of pandemic strains of HIV and will apply to other viruses as well. To address this question, SIV strains obtained directly from sooty mangabeys are being used to infect rhesus macaques to understand how SIV adapts to a new host species. The results of these studies will contribute to the understanding of how new epidemic strains of HIV evolved and emerged in the 20th century. It is well known that HIV infection of humans is fatal in five to ten years if left untreated. However, a small percentage of HIV infected persons suppress the infection naturally and live for longer periods without symptoms of AIDS. Little is known about these elite long-term nonprogressors (LTNP), and it is difficult to study in humans, especially in the early stages of infection. Research in the Division has recently shown that a significant percentage of SIVmac-infected rhesus of Chinese origin become LTNP analgous to elite LTNP in humans. Research is ongoing to understand the dynamics of immune cell restoration and control of infection in these animals. Several species of nonhuman primates native to Africa are naturally infected with SIV and rarely develop disease. Understanding how these animals can maintain a life-long infection without resulting in disease is likely to offer important clues to help develop strategies to prevent AIDS in humans. Toward this end, we are examining African green monkeys and sooty mangabeys. Each species is infected with its own strain of SIV, and each seems to have developed unique mechanisms to cope with the infection. Only two examples of AIDS are known in naturally infected sooty mangabeys and African green monkeys. Both studies were done in the TNPRC Division of Microbiology.
Heterosexual Models for HIV Transmission
The majority of HIV infections occur by heterosexual transmission. Investigators in the Division of Microbiology were the first to develop an animal model to study sexual transmission. Using SIVmac and female rhesus macaques, infection was readily transmitted across the intact vaginal lining. Infection became systemic and resulted in the development of AIDS. This model closely follows AIDS development in HIV-infected women and is being used to study prevention, treatment and pathogenesis. Recent studies in collaboration with the Division of Comparative Pathology have focused on monoclonal antibodies to prevent SIV vaginal infection. There is a critical need for inexpensive drugs that can prevent HIV transmission to women because vaccines are not yet ready for use. Drugs that can be used safely and discreetly by women to prevent HIV infection are of great interest, particularly in developing countries. The monoclonal antibody b12 had been used as a topical prophylactic in the vagina and shown to be effective at blocking infection. However, b12 is rare among antibodies in that it can neutralize HIV and SHIV (a simian human hybrid virus). In ongoing studies, mutants of b12 are being tested to understand the mechanism by which b12 antibody can prevent vaginal SHIV infection.
Other uses of the model are the study of the role of the female hormones, progesterone and estrogen, in SIV vaginal transmission. A long-term study has shown that progesterone increases the susceptibility of the vaginal lining to SIV infection. In contrast, estrogen prevents infection. Prevention correlated with an increase in thickness of the vaginal lining which is hypothesized to block infection. This finding can be exploited to develop new prevention strategies. The most recent development applies a vaginal cream containing estrial. Estrogen-based vaginal creams are routinely used by women to treat post-menopausal vaginal symptoms. Estrogen creams induce changes in the vaginal microenvironment that increase resistance to SIV infection. The results have been promising, and a new study is planned in women to examine the changes in the vagina that may promote resistance to HIV infection.
AIDS vaccines
AIDS vaccines are a major focus in the Division of Microbiology. Live vectors and immunogenic peptides have been used. The SIV macaque model is also used by Division collaborators having AIDS vaccine programs funded by NIH.
Live vaccine vectors have included vesicular stomatitis virus (VSV). VSV is relatively non-pathogenic in monkeys and humans. VSV-HIV hybrid viruses have been constructed to express HIV and SIV genes. The safety, immunogenicity and efficacy of these vectors expressing SIV genes have been tested in the SIV/macaque model of AIDS. Animals have been immunized by intramuscular, oral and intranasal routes to determine which induces the strongest and most durable immune response. The humoral and cell-mediated immune responses were measured. The appearance of antibodies was measured and compared between different VSV-hybrids and different routes of immunization. SHIV was used to challenge immunized macaque monkeys. The candidate vaccine induced strong protection in the SHIV model. The immunized monkeys were not observed to have any deleterious effects from the immunization protocol with the live vectors. Upon challenge with a pathogenic SHIV virus, the immunized monkeys suppressed the virus compared to the mock vaccine control group. Because of the success of this series of experiments,VSV-HIV hybrid vectors are being developed for clinical trials in humans. These new vectors for human use are currently being tested in rhesus macaques to determine if they are safe and immunogenic.
Another vaccine approach in the Division of Microbiology is the use of synthetic peptides derived from immunogenic epitopes of the gag and env gene of HIV and SIV. These peptides were incorporated into immunostimulating complexes known as ISCOMS. This and other novel approaches are being pursued in the Division of Microbiology.
 |
 |
